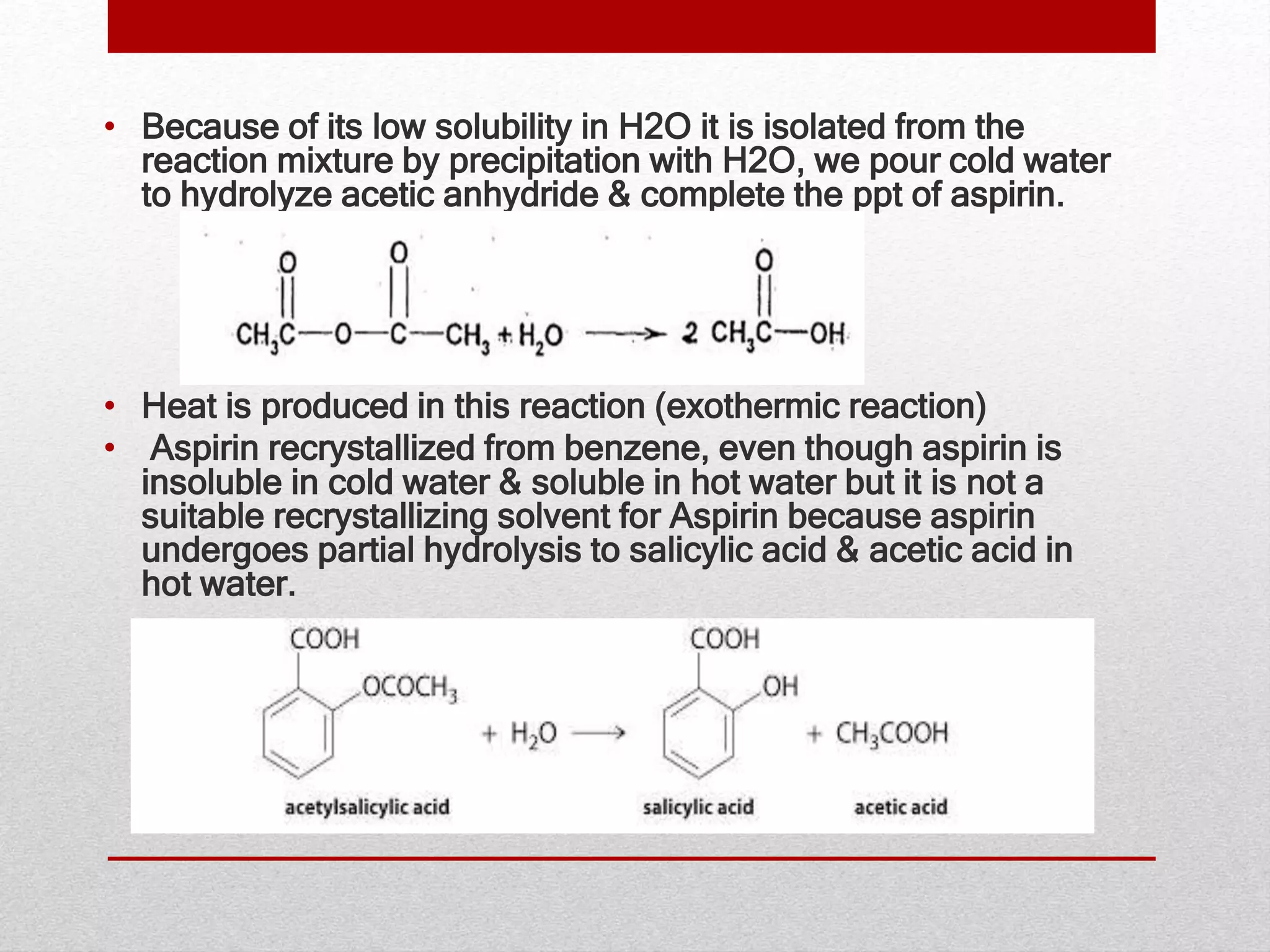
Aspirin, a widely used medicine known for its pain relieving, anti inflammatory, and antipyretic properties, has a fascinating history and chemical background. The Aspirin Preparation Reaction, which involves the synthesis of aspirin from salicylic acid and acetic anhydride, is a classic exemplar of an esterification reaction. This process not only highlights the chemical principles behind aspirin but also provides insights into the industrial product of this essential drug.
Historical Background of Aspirin
Aspirin s journey began with the ancient use of willow bark, which contains salicin, a harbinger to salicylic acid. The fighting ingredient in willow bark was sequestrate and purify in the 19th century, prima to the development of salicylic acid. However, salicylic acid was found to be too harsh on the stomach, cue the search for a safer alternative. In 1897, Felix Hoffmann, a chemist act for Bayer, successfully synthesize acetylsalicylic acid, which we now cognize as aspirin. This discovery revolutionized the treatment of pain and fervour, do aspirin one of the most commonly used medications worldwide.
The Chemistry Behind Aspirin Preparation Reaction
The Aspirin Preparation Reaction involves the response between salicylic acid and acetic anhydride to form acetylsalicylic acid (aspirin) and acetic acid as a byproduct. This response is a classic example of an esterification procedure, where an acid reacts with an alcohol to form an ester and water. In this case, the carboxyl acid group of salicylic acid reacts with the anhydride to form the ester, aspirin.
The chemic equation for the Aspirin Preparation Reaction is as follows:

Step by Step Guide to Aspirin Preparation Reaction
Conducting the Aspirin Preparation Reaction in a laboratory determine involves respective steps. Here is a detail guidebook to execute this reaction:
Materials Needed
- Salicylic acid
- Acetic anhydride
- Sulfuric acid (catalyst)
- Distilled water
- Ethanol
- Beaker
- Stirring rod
- Hot plate
- Filter newspaper
- Vacuum desiccator
Procedure
1. Preparation of Reagents: Measure out the demand amounts of salicylic acid and acetic anhydride. Typically, a 1: 1 molar ratio is used.
2. Addition of Catalyst: Add a few drops of sulphuric acid to the motley. Sulfuric acid acts as a catalyst, speeding up the response.
3. Heating the Mixture: Place the beaker contain the assortment on a hot plate and heat mildly. Stir the miscellanea continuously to ensure even heating.
4. Formation of Aspirin: As the intermixture heats, the response between salicylic acid and acetic anhydride will occur, forming acetylsalicylic acid (aspirin) and acetic acid. The reaction is exothermic, so careful temperature control is indispensable.
5. Cooling and Crystallization: Allow the motley to cool to room temperature. As it cools, aspirin will commence to crystallize out of the result.
6. Filtration: Filter the mixture using filter newspaper to secern the solid aspirin crystals from the liquid.
7. Washing: Wash the crystals with cold distilled water to remove any rest acetic acid and sulfuric acid.
8. Drying: Place the washed crystals in a vacuum desiccator to dry altogether. This step ensures that any residual moisture is take, leave pure aspirin.
Note: Always treat chemicals with care, using appropriate personal protective equipment (PPE) such as gloves and safety glasses. Perform the response in a well vent country or under a fume hood to avoid inhale harmful fumes.
Mechanism of the Aspirin Preparation Reaction
The mechanism of the Aspirin Preparation Reaction involves respective steps:
- Protonation: The carboxylic acid group of salicylic acid is protonated by the sulfuric acid catalyst, create it more reactive.
- Nucleophilic Attack: The protonated carboxyl acid group attacks the carbonyl carbon of the acetic anhydride, forming a tetrahedral intermediate.
- Collapse of Intermediate: The tetrahedral mediate collapses, turn acetic acid and organize the ester bond between the salicylic acid and the acetyl group.
- Deprotonation: The resulting protonated ester is deprotonated, forming acetylsalicylic acid (aspirin).
Applications and Importance of Aspirin
Aspirin has a wide range of applications in medicine, do it one of the most versatile drugs available. Some of its key uses include:
- Pain Relief: Aspirin is commonly used to relieve mild to moderate pain, including headaches, muscle aches, and arthritis.
- Anti Inflammatory: It reduces inflammation, get it effective in treating conditions like rheumatoid arthritis and other inflammatory disorders.
- Antipyretic: Aspirin lowers pyrexia by conquer the product of prostaglandins, which are affect in the body s temperature regulation.
- Cardiovascular Health: Low doses of aspirin are oftentimes order to prevent heart attacks and strokes by conquer platelet collection.
Safety and Precautions
While aspirin is loosely safe when used as directed, it is important to be aware of potential side effects and precautions. Some key points to consider include:
- Gastrointestinal Issues: Aspirin can cause stomach upset, ulcers, and hemorrhage, specially when taken in eminent doses or for prolonged periods.
- Allergic Reactions: Some individuals may be allergic to aspirin, see symptoms such as hives, tumefy, and difficulty breathing.
- Interactions with Other Medications: Aspirin can interact with other medications, include blood thinners, nonsteroidal anti inflammatory drugs (NSAIDs), and certain antidepressants.
- Contraindications: Aspirin should be avoided by individuals with certain medical conditions, such as bleeding disorders, ulcers, and severe liver or kidney disease.
Always consult a healthcare supplier before commence any new medicine, including aspirin, to assure it is safe and appropriate for your item-by-item health needs.
Note: Aspirin should not be given to children or teenagers with viral infections, as it has been linked to a rare but dangerous status telephone Reye's syndrome.
Future Directions in Aspirin Research
The Aspirin Preparation Reaction and the drug itself preserve to be areas of fighting research. Scientists are explore new ways to synthesize aspirin more expeditiously and sustainably, as easily as investigating its possible applications in other medical fields. Some emerging areas of research include:
- Nanotechnology: Researchers are developing nanoscale aspirin particles that can be targeted to specific tissues or cells, heighten its therapeutical effects and trim side effects.
- Combination Therapies: Combining aspirin with other drugs or therapies to raise its effectuality in process conditions like cancer and neurodegenerative diseases.
- Personalized Medicine: Tailoring aspirin doses and formulations to single patients free-base on their genic makeup and health status.
As our realize of aspirin's mechanisms of action and possible applications continues to grow, so too will its role in mod medicine.
to summarise, the Aspirin Preparation Reaction is a key process in the synthesis of one of the existence s most widely used medications. From its historical roots to its modern applications, aspirin continues to play a crucial role in healthcare. Understanding the chemistry behind its preparation not only provides insights into its sanative properties but also highlights the importance of chemical reactions in the development of life saving drugs. The ongoing research into aspirin s likely applications ensures that it will remain a cornerstone of medical treatment for years to come.
Related Terms:
- skin rash induce by aspirin
- aspirin reaction formula
- aspirin allergy symptoms mayo clinic
- aspirin allergic response
- can aspirin hurt your stomach
- aspirin side effects