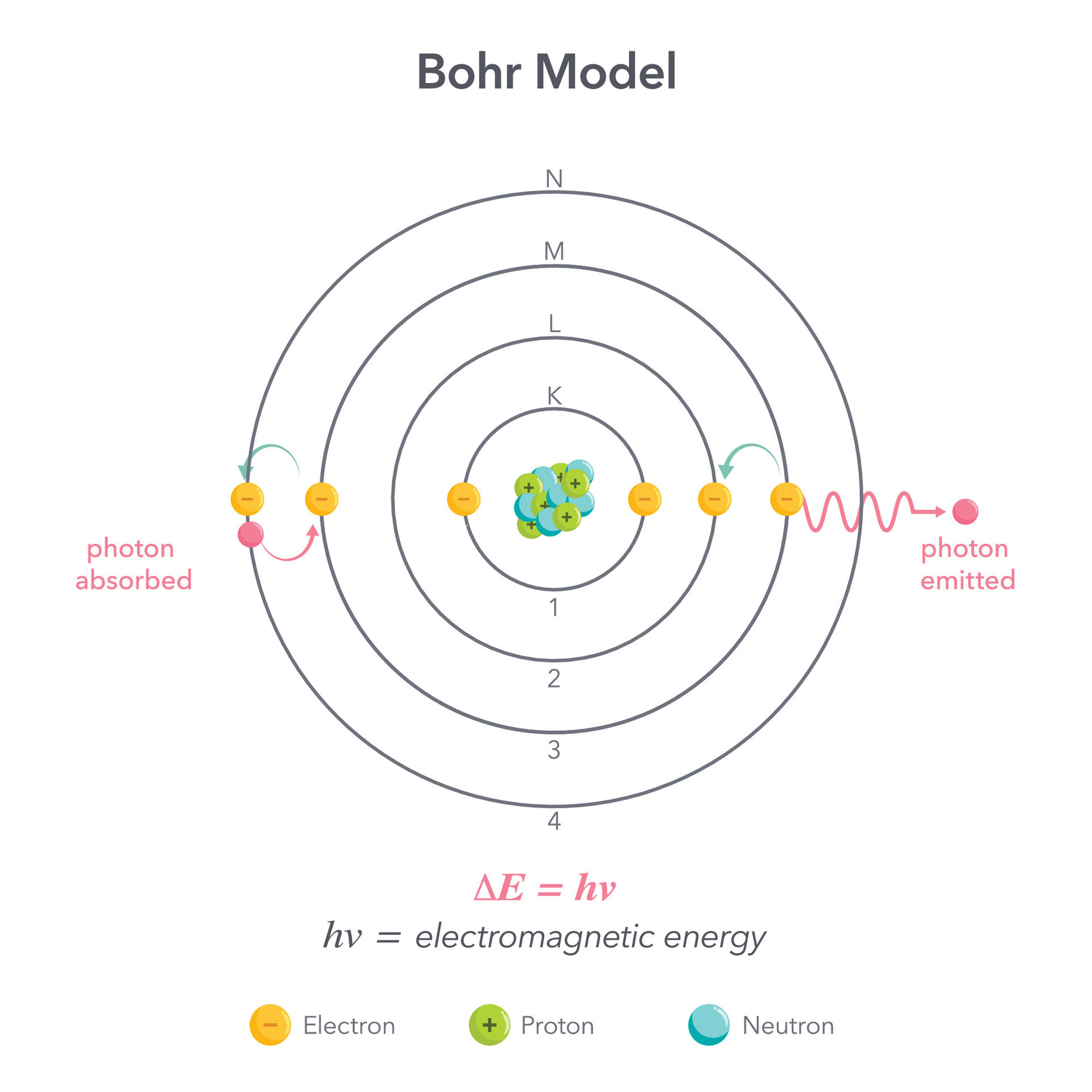
The Bohr Model of Helium is a fundamental concept in the study of atomic construction and quantum mechanics. Developed by Niels Bohr in 1913, this model provides a simplified representation of the helium atom, which consists of two protons and two neutrons in the nucleus, skirt by two electrons. The Bohr Model of Helium is particularly useful for understanding the basic principles of electron configuration and energy levels within atoms.
The Basics of the Bohr Model
The Bohr Model of Helium is based on several key principles that account the conduct of electrons within an atom. These principles include:
- Electrons orbit the nucleus in bushel, circular paths called orbits or energy levels.
- Each orbit corresponds to a specific energy level, with the lowest energy tier being closest to the nucleus.
- Electrons can jump between energy levels by assimilate or breathe specific amounts of energy, typically in the form of photons.
- The angular momentum of an electron in a give orbit is quantized, meaning it can only direct on specific discrete values.
Electron Configuration in the Bohr Model of Helium
The Bohr Model of Helium helps illustrate the electron configuration of the helium atom. Helium has an atomic number of 2, which means it has two protons in its nucleus and two electrons orbiting around it. According to the Bohr Model, these two electrons occupy the first energy level, which is the closest to the nucleus.
In the Bohr Model of Helium, the electron configuration is symbolize as 1s 2. This notation indicates that both electrons are in the first energy level (n 1) and the s subshell. The superscript 2 denotes that there are two electrons in this subshell.
Energy Levels and Quantum Numbers
The Bohr Model of Helium introduces the concept of energy levels and quantum numbers, which are essential for understanding the behavior of electrons within an atom. The master quantum number (n) determines the energy level of an electron. For helium, the two electrons are in the first energy stage (n 1).
Additionally, the Bohr Model of Helium considers the angular momentum quantum number (l), which describes the shape of the electron s orbit. For the first energy point, l can only be 0, agree to an s orbital. The magnetized quantum bit (m l ) and spin quantum number (ms ) further describe the orientation and spin of the electron, respectively.
Stability and Inertness of Helium
The Bohr Model of Helium explains why helium is a stable and inert gas. With its two electrons entirely occupy the first energy stage, helium has a full valency shell. This configuration makes helium extremely stable and tolerant to chemic reactions. The constancy of helium is a direct result of its electron configuration, which is absolutely equilibrize according to the Bohr Model.
Limitations of the Bohr Model
While the Bohr Model of Helium is a valuable puppet for see the canonical structure of the helium atom, it has various limitations. These limitations include:
- It does not accurately draw the behavior of electrons in atoms with more than one electron.
- It does not account for the wave like properties of electrons, which are punter described by quantum mechanics.
- It does not explicate the fine construction of apparitional lines, which arises from the interaction between the electron s spin and orbital angular momentum.
Despite these limitations, the Bohr Model of Helium remains a utile conceptual framework for introducing the principles of nuclear structure and electron shape.
Applications of the Bohr Model
The Bohr Model of Helium has several virtual applications in assorted fields of science and engineering. Some of these applications include:
- Education: The Bohr Model of Helium is wide used in educational settings to teach students about atomic structure, electron conformation, and energy levels.
- Spectroscopy: The model helps excuse the emission and absorption spectra of atoms, which are used in spectroscopy to identify elements and study their properties.
- Chemistry: Understanding the Bohr Model of Helium is crucial for comprehending the chemical demeanor of elements, include their reactivity and adhere properties.
- Physics: The model provides a foundation for more progress theories in quantum mechanics and atomic physics.
Comparing the Bohr Model with Modern Atomic Models
The Bohr Model of Helium, while historically substantial, has been superseded by more advance atomic models that provide a more accurate description of nuclear structure. These modern models include:
- Quantum Mechanical Model: This model describes electrons as wave functions rather than particles, cater a more accurate representation of electron behavior.
- Schrödinger Model: Based on the Schrödinger equivalence, this model predicts the probability dispersion of electrons within an atom.
- Orbital Model: This model describes electrons in terms of orbitals, which are regions of space where electrons are probable to be found.
While these mod models proffer a more comprehensive understanding of nuclear structure, the Bohr Model of Helium remains a worthful instrument for introductory purposes.
Helium s Role in the Periodic Table
Helium s view in the occasional table is now related to its electron constellation as describe by the Bohr Model of Helium. Helium is the first element in the noble gas group (Group 18), which includes elements with full valence shells. This full valence shell makes helium extremely stable and non reactive, a characteristic share by all noble gases.
In the occasional table, helium is lay in the first period and the 18th group. Its atomic routine is 2, and its electron configuration is 1s 2. This shape is all-important for interpret helium s chemic properties and its role in the periodic table.
Helium s Unique Properties
Helium s unique properties can be understood through the lens of the Bohr Model of Helium. Some of these properties include:
- Low Boiling Point: Helium has the lowest boil point of any element, at roughly 268. 9 C. This is due to its weak intermolecular forces, which result from its full valency shell and lack of chemical reactivity.
- Low Density: Helium is the second lightest element, with a concentration of 0. 1785 g L at standard temperature and pressure. This low density makes it utilitarian in applications such as airships and balloons.
- Non Reactiveness: Helium is chemically inert, meaning it does not react with other elements under normal conditions. This inertness is a unmediated result of its full valence shell, as draw by the Bohr Model of Helium.
Helium in Everyday Life
Helium s unparalleled properties create it valuable in several everyday applications. Some of these applications include:
- Medical Use: Helium is used in medical project techniques such as magnetised plangency imaging (MRI) and in respiratory treatments for conditions like asthma.
- Industrial Use: Helium is used in weld, where it provides an inert atmosphere to prevent oxidation. It is also used in the production of semiconductors and fiber optics.
- Scientific Research: Helium is used in cryogenics to accomplish super low temperatures, which are necessary for certain scientific experiments and technologies.
- Consumer Products: Helium is ordinarily used to fill balloons and airships, ply lift due to its low concentration.
Helium s Role in the Universe
Helium is the second most abundant element in the universe, after hydrogen. It plays a crucial role in the formation of stars and galaxies. The Bohr Model of Helium helps explain the nuclear fusion processes that occur in stars, where hydrogen atoms combine to form helium, unloosen tremendous amounts of energy.
In the betimes universe, helium was formed during the Big Bang nucleosynthesis, a process that occurred within the first few minutes after the Big Bang. This summons produced most the helium present in the universe today.
Helium s Isotopes
Helium has respective isotopes, but only two are stable: helium 3 and helium 4. The Bohr Model of Helium can be utilise to these isotopes to read their nuclear structure. Helium 4 is the most abundant isotope, lie of two protons and two neutrons in the nucleus. Helium 3, conversely, has one fewer neutron.
The table below summarizes the properties of helium s stable isotopes:
| Isotope | Number of Protons | Number of Neutrons | Abundance |
|---|---|---|---|
| Helium 3 | 2 | 1 | 0. 000137 |
| Helium 4 | 2 | 2 | 99. 999863 |
Note: The abundance of helium 3 is extremely low compared to helium 4, create helium 4 the prevailing isotope in natural helium.
Helium s Discovery and History
Helium was first discover in 1868 by French stargazer Pierre Janssen and English stargazer Norman Lockyer. They observed a yellow spectral line in the sun s spectrum that did not correspond to any known element. This line was later identified as helium, derived from the Greek word helios, signify sun.
It was not until 1895 that helium was first insulate on Earth by Scottish druggist William Ramsay. Ramsay extracted helium from the mineral cleveite, a uranium bearing mineral. This discovery sustain the world of helium on Earth and pave the way for further research into its properties and applications.
In 1908, helium was first liquefied by Dutch physicist Heike Kamerlingh Onnes, who achieved a temperature of 4. 2 K (268. 9 C). This breakthrough was all-important for the development of cryogenics and the study of low temperature physics.
In the 20th century, helium's unique properties made it worthful for assorted applications, include airships, weld, and scientific inquiry. During World War II, helium was used in military applications, such as blimps and balloons, to detect enemy submarines.
Today, helium is chiefly extracted from natural gas reserves, where it is found in small quantities. The United States is the world's largest manufacturer of helium, with important reserves in Texas, Oklahoma, and Kansas.
Helium's scarcity and increase demand have led to concerns about its availability in the future. Efforts are being made to acquire new technologies for helium origin and reuse to secure a sustainable supply.
In recent years, the study of helium has expanded to include its role in quantum mechanics and superconductivity. Helium 4, in particular, exhibits unique properties at low temperatures, such as superfluidity, where it flows without viscosity. This phenomenon has important implications for the study of quantum fluids and the development of new technologies.
Helium's role in the Bohr Model of Helium provides a fundament for realise its nuclear construction and properties. By canvass helium, scientists have gained insights into the behavior of electrons, the nature of nuclear nuclei, and the primal principles of quantum mechanics.
Helium's unique properties and applications create it a valuable element in respective fields, from medicine and industry to scientific enquiry and consumer products. Its role in the universe and its discovery have contributed to our understanding of the cosmos and the elements that compose it.
Helium s journey from its discovery in the sun to its widespread use on Earth is a testament to the power of scientific inquiry and the importance of understanding the underlying building blocks of the universe. The Bohr Model of Helium, while a simplify representation, continues to inspire and educate, supply a gateway to the intrigue world of atomic structure and quantum mechanics.
Related Terms:
- bohr model diagram model
- bohr model
- helium bohr rutherford diagram
- bohr model chart
- bohr model excuse
- helium bohr model diagram