Understanding the def freezing point of h2o is rudimentary to various scientific and industrial processes. The freezing point of h2o is the temperature at which liquid water transitions into solid ice. This conversion is crucial in many fields, including meteorology, chemistry, and organize. This blog post will delve into the science behind the def freeze point, its implication, and its applications in different industries.
What is the Def Freezing Point?
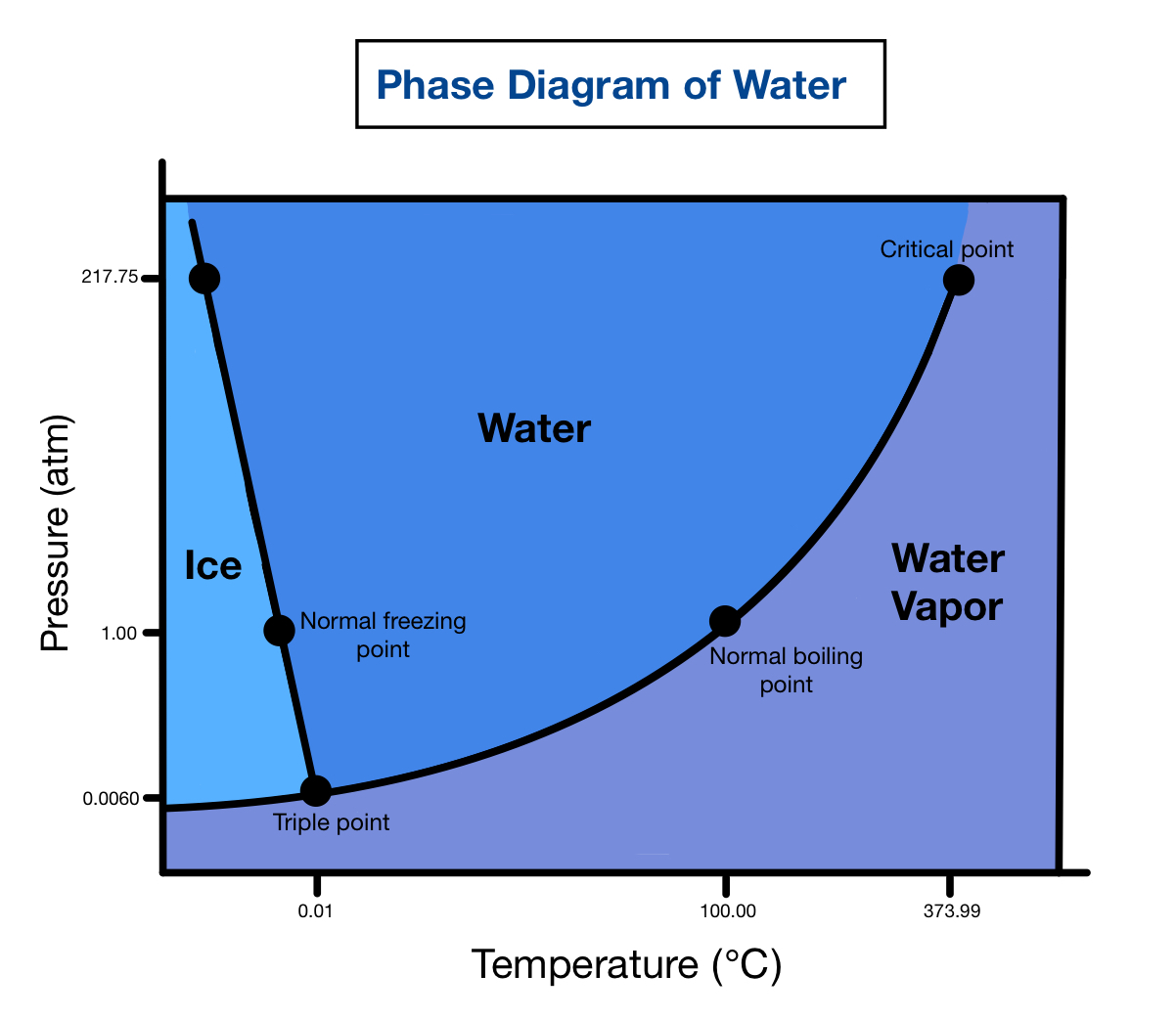
The def freezing point of h2o is 0 degrees Celsius (32 degrees Fahrenheit) under standard atmospheric conditions. This means that at this temperature, h2o molecules slow down enough to form a solid structure, leave in ice. The freeze point can vary slimly depending on factors such as pressing and the front of impurities. For case, adding salt to water lowers its freeze point, a phenomenon known as freezing point depression.
Factors Affecting the Def Freezing Point
Several factors can influence the def freezing point of h2o. Understanding these factors is all-important for various applications:
- Pressure: Increasing pressure can lower the freeze point of water. This is why ice can form at temperatures below 0 C under high pressure.
- Impurities: The front of impurities, such as salt or other solutes, can lower the freezing point. This is why brine freezes at a lower temperature than pure water.
- Volume: The volume of water can also affect its freeze point. Larger volumes of water may freeze at slimly different temperatures due to differences in heat distribution.
Applications of the Def Freezing Point
The def freeze point of h2o has legion applications across various industries. Here are some key areas where translate the freeze point is all-important:
Meteorology
In meteorology, the def freezing point is essential for foreshadow conditions patterns. Meteorologists use this info to forecast the formation of ice, snow, and other downfall types. Understanding the freezing point helps in issue warnings for icy roads and other conditions pertain hazards.
Chemistry
In chemistry, the def freezing point is used in various experiments and processes. for instance, freeze point depression is a mutual technique used to determine the molecular weight of solutes in a resolution. This method involves mensurate the dispute in freezing points between pure h2o and a solvent containing the solute.
Engineering
In engineer, the def freeze point is all-important for designing systems that work in cold environments. Engineers must consider the freeze point when designing pipelines, bridges, and other structures to ensure they can withstand low temperatures without damage. For illustration, in cold regions, engineers use antifreeze solutions to prevent h2o from freeze in pipes and engines.
Food Industry
In the food industry, the def freeze point is important for maintain food. Freezing is a mutual method used to extend the shelf life of perishable items. Understanding the freeze point helps in influence the optimum temperature for store and enthral food products. for representative, ice cream manufacturers must carefully control the freezing summons to attain the hope texture and consistency.
Agriculture
In husbandry, the def freezing point is crucial for protecting crops from frost damage. Farmers use various techniques, such as continue crops with pliant sheets or using irrigation systems, to prevent freeze temperatures from harming their crops. Understanding the freeze point helps farmers make informed decisions about when to harvest and how to protect their crops.
Measuring the Def Freezing Point
Measuring the def freeze point of h2o involves precise instruments and techniques. Here are some common methods used to mensurate the freeze point:
- Thermometers: Digital thermometers are commonly used to measure the freezing point. These devices provide accurate readings and are easy to use.
- Differential Scanning Calorimetry (DSC): DSC is a technique used to mensurate the heat flow consociate with phase transitions. It provides detailed information about the freezing point and other caloric properties of a nitty-gritty.
- Freezing Point Depression: This method involves measuring the departure in freeze points between pure water and a solvent bear a solute. It is ordinarily used in chemistry to determine the molecular weight of solutes.
Note: When measuring the def freezing point, it is crucial to guarantee that the sample is pure and free from impurities. Impurities can affect the accuracy of the measurement.
Importance of the Def Freezing Point in Everyday Life
The def freezing point of water plays a substantial role in our daily lives. Here are some examples of how it affects us:
- Driving Conditions: Understanding the freezing point helps drivers prepare for icy roads and other wintertime driving conditions. Knowing when to expect ice formation can prevent accidents and ensure safe travel.
- Home Maintenance: Homeowners must deal the freeze point when winterise their homes. This includes isolate pipes, drain h2o from outdoor faucets, and using antifreeze solutions to prevent damage from freezing temperatures.
- Sports and Recreation: Athletes and outdoor enthusiasts must be aware of the freeze point when enter in wintertime sports. Knowing when to expect ice establishment can help prevent injuries and ensure a safe and enjoyable experience.
The Science Behind the Def Freezing Point
The def freezing point of h2o is determined by the molecular construction and interactions of h2o molecules. Water molecules are polar, meaning they have a positive and negative end. This sign allows water molecules to form hydrogen bonds with each other. As the temperature decreases, the energizing energy of the h2o molecules decreases, and they slow down. At the freezing point, the molecules arrange themselves into a crystalline structure, constitute ice.
The presence of impurities can disrupt the establishment of hydrogen bonds, lower the freezing point. This is why adding salt to h2o lowers its freeze point. The salt ions interfere with the hydrogen bind between h2o molecules, get it harder for them to form a solid construction.
The def freeze point of h2o is a fundamental concept in science and has legion applications in diverse industries. Understanding this concept is all-important for predicting weather patterns, plan direct systems, preserving food, and protect crops. By measure and command the freezing point, we can ensure the safety and efficiency of many processes and systems.
to summarize, the def freeze point of h2o is a critical concept with across-the-board swan implications. From meteorology to organize, and from chemistry to usda, see the freeze point is essential for various applications. By measuring and command the freeze point, we can ensure the safety and efficiency of many processes and systems, making it a key concept in science and industry.
Related Terms:
- def fluid antifreeze additive
- def fluid freeze point
- what keeps def from freeze
- def freezes at what temp
- does def fluid freeze
- def fluid freeze point