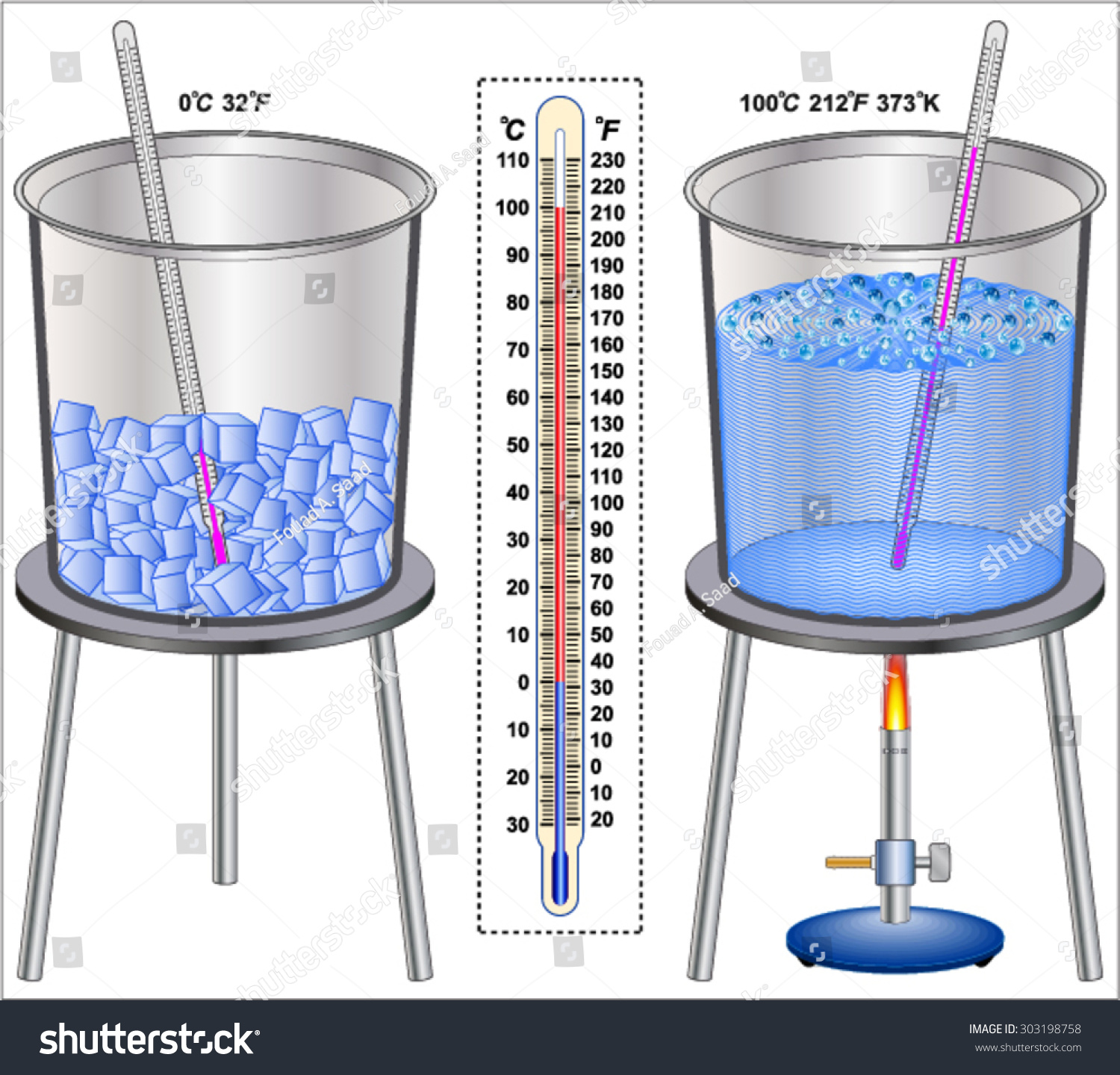
Understanding the melting point of ice is rudimentary to assorted scientific and practical applications. The melting point of ice, which is 0 C (32 F) at standard atmospheric pressing, is a critical concept in fields ranging from meteorology to materials skill. This blog post delves into the science behind the unthaw point of ice, its significance, and its applications in different domains.
The Science Behind the Melting Point of Ice
The melting point of ice is the temperature at which solid water (ice) transitions to liquid h2o. This phase transition is rule by the principles of thermodynamics and molecular interactions. At the mellow point, the thermal energy of the molecules in ice is sufficient to overcome the intermolecular forces that hold the water molecules in a rigid crystal structure. As a consequence, the molecules gain enough kinetic energy to move freely, metamorphose the ice into liquid h2o.
Several factors influence the dethaw point of ice:
- Pressure: Increasing pressing can lower the unfreeze point of ice. This phenomenon is known as the press run point and is important in the formation of glaciers and ice skating.
- Impurities: The front of impurities, such as salt or other solutes, can lower the melting point of ice. This is why salt is used to melt ice on roads during wintertime.
- Temperature: The melting point of ice is straight involve by temperature. At standard atmospherical pressing, the thaw point is 0 C (32 F).
Significance of the Melting Point of Ice
The melting point of ice has profound implications in diverse scientific and practical contexts. Understanding this concept is essential for:
- Climate Science: The dissolve point of ice plays a crucial role in climate studies. Changes in global temperatures can touch the melting of polar ice caps and glaciers, starring to sea point rise and other climatic changes.
- Meteorology: Weather predict relies on translate the run point of ice to predict downfall types, such as rain, snow, or sleet.
- Materials Science: The study of phase transitions, including the thaw point of ice, is lively in materials science for developing new materials with specific properties.
- Engineering: In civil and mechanical mastermind, understanding the mellow point of ice is crucial for project structures that can withstand freeze temperatures and for developing de icing systems.
Applications of the Melting Point of Ice
The dissolve point of ice has numerous applications across different fields. Some of the key applications include:
- Road Safety: Salt and other de icing agents are used to lower the run point of ice on roads, making them safer for driving during wintertime.
- Food Preservation: The mellow point of ice is important in food saving techniques, such as freezing, to maintain the caliber and novelty of perishable items.
- Industrial Processes: In industries such as chemic manufacturing and metallurgy, the run point of ice is used to control temperatures and phase transitions in diverse processes.
- Environmental Monitoring: Understanding the melting point of ice is essential for supervise environmental changes, such as the melting of glaciers and polar ice caps, which can signal climate alter.
Experimental Determination of the Melting Point of Ice
Determining the melting point of ice experimentally involves various steps. Here is a detailed routine:
1. Prepare the Ice Sample: Obtain a pure sample of ice. Ensure that the ice is free from impurities to get an accurate thaw point.
2. Set Up the Apparatus: Use a thermometer and a beaker or test tube to hold the ice sample. Place the apparatus in a operate environment, such as a h2o bath or an isolate container.
3. Heat the Sample: Gradually heat the ice sample using a heat source, such as a Bunsen burner or a hot plate. Monitor the temperature using the thermometer.
4. Observe the Phase Transition: Note the temperature at which the ice begins to melt and becomes liquid water. This temperature is the melting point of ice.
5. Record the Data: Record the observed melting point and any other relevant information, such as the rate of inflame and the honour of the ice sample.
Note: Ensure that the heating summons is gradual and controlled to avoid superheating or supercooling, which can impact the accuracy of the melting point determination.
Factors Affecting the Melting Point of Ice
Several factors can influence the melting point of ice. Understanding these factors is crucial for accurate measurements and applications. Some of the key factors include:
- Pressure: As mention earlier, increase pressure can lower the melting point of ice. This is why ice skates can glide smoothly on ice the pressure from the skates lowers the melting point, creating a thin layer of h2o.
- Impurities: The front of impurities, such as salt or other solutes, can lower the melt point of ice. This is known as freeze point slump and is used in de icing applications.
- Temperature: The melting point of ice is straight affect by temperature. At standard atmospheric pressing, the melting point is 0 C (32 F).
Here is a table summarizing the effects of different factors on the dethaw point of ice:
| Factor | Effect on Melting Point |
|---|---|
| Pressure | Increases press lowers the melting point |
| Impurities | Presence of impurities lowers the melt point |
| Temperature | Directly affects the melting point |
Real World Examples of the Melting Point of Ice
The dissolve point of ice has numerous real world applications. Here are some examples:
- Ice Skating: The pressing from ice skates lowers the dissolve point of ice, make a thin stratum of h2o that allows skaters to glide swimmingly.
- De Icing Roads: Salt and other de icing agents are used to lower the run point of ice on roads, get them safer for motor during winter.
- Food Preservation: The melting point of ice is crucial in food preservation techniques, such as freezing, to maintain the lineament and freshness of perishable items.
- Climate Change Monitoring: Understanding the melting point of ice is indispensable for monitor environmental changes, such as the melting of glaciers and polar ice caps, which can indicate climate change.
These examples illustrate the pragmatic implication of the unthaw point of ice in respective domains.
to summarize, the melting point of ice is a fundamental concept with wide ranging applications. Understanding the skill behind it, its implication, and its applications is essential for respective fields, from climate science to materials orchestrate. By search the factors that influence the dethaw point of ice and its real creation examples, we gain a deeper grasp for this crucial scientific principle.
Related Terms:
- ice melt temperature chart
- melting point vs temperature
- where is the dethaw point
- ice melts at what temperature
- ice melts into liquid h2o
- when ice melts its temperature